What is binding energy per nucleon and why is it important?
Binding energy per nucleon is the average amount of energy required to remove a single proton or neutron from a nucleus. It is calculated by dividing the total nuclear binding energy by the total number of nucleons (protons + neutrons). This value provides a useful way to compare the relative stability of different nuclei.
Binding energy per nucleon measures on average how tightly each nucleon is bound within a nucleus and is a key indicator of nuclear stability.
Nuclei with a higher binding energy per nucleon are generally more stable because each nucleon is more tightly bound within the nucleus. Conversely, nuclei with lower binding energy per nucleon are more likely to undergo radioactive decay in order to move toward a more stable configuration.
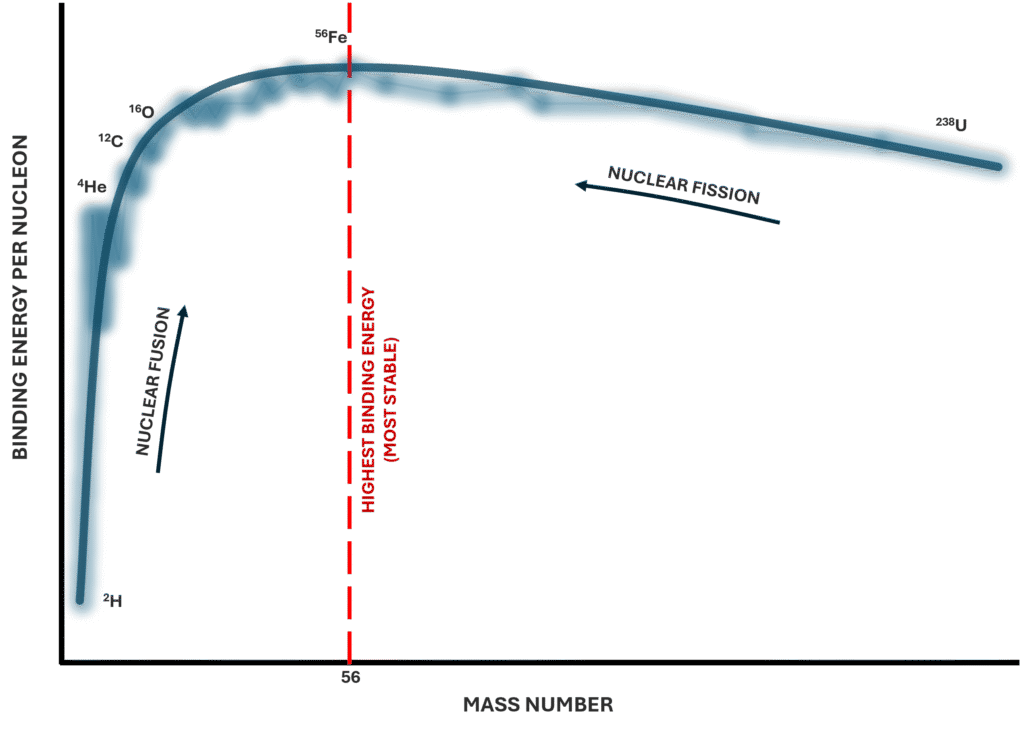
The variation of binding energy per nucleon across the periodic table explains why very light nuclei can release energy through fusion, and very heavy nuclei can release energy through fission. It also explains why certain nuclei, such as iron-56, are particularly stable.
Understanding the physics
Total binding energy increases as nuclei become larger, but this does not necessarily mean larger nuclei are more stable. What matters is how tightly each nucleon is bound on average.
When binding energy per nucleon is plotted against atomic mass number, the curve rises sharply for light nuclei, reaches a peak around iron-56, and then gradually declines for heavier nuclei. This peak represents the most energetically favourable configuration of nucleons.
For light nuclei, adding nucleons increases stability because more strong-force interactions occur between neighbouring nucleons. The strong nuclear force is short-range, so each nucleon interacts mainly with its immediate neighbours. As more nucleons are added (up to a point), the total attractive interactions increase and binding energy per nucleon rises.
However, as nuclei become very large, proton–proton electrostatic repulsion becomes increasingly significant. Although neutrons help offset this repulsion by contributing strong-force attraction without adding charge, beyond a certain size the repulsive forces reduce overall stability. This causes binding energy per nucleon to fall in heavy nuclei.
Radioactive decay tends to occur when a nucleus can move toward a configuration with higher binding energy per nucleon. In other words, decay is energetically favourable if it increases the average stability of the nucleons.
Where this matters clinically
Binding energy per nucleon explains why radionuclides used in nuclear medicine release energy when they decay. The emitted radiation ultimately arises because the daughter nucleus has a more stable (higher binding energy per nucleon) configuration than the parent. Understanding this principle clarifies why unstable isotopes exist at all, and why they can be used for imaging.
