What is nuclear binding energy?
Nuclear binding energy is the energy required to completely separate a nucleus into its individual protons and neutrons. It represents how strongly the nucleons are held together by the strong nuclear force. The greater the binding energy, the more stable the nucleus.
Nuclear binding energy is the energy required to separate a nucleus into its nucleons and reflects how strongly the nucleus is held together.
Binding energy arises because the total mass of a bound nucleus is slightly less than the combined mass of its individual protons and neutrons. This “missing” mass has been converted into energy according to Einstein’s equation:
E = mc2
That energy is the binding energy holding the nucleus together.
Nuclei with high binding energy are more resistant to radioactive decay, while those with lower binding energy are more likely to be unstable.
Understanding the physics
When protons and neutrons come together to form a nucleus, energy is released as the strong nuclear force binds them into a lower-energy configuration. Because energy and mass are interchangeable, this released energy corresponds to a reduction in mass. The difference between the summed masses of free nucleons and the actual mass of the nucleus is called the mass defect.
Mathematically:
Binding energy = (mass defect) × c2
This binding energy reflects how much energy would need to be supplied to overcome the strong nuclear force and pull the nucleus apart.
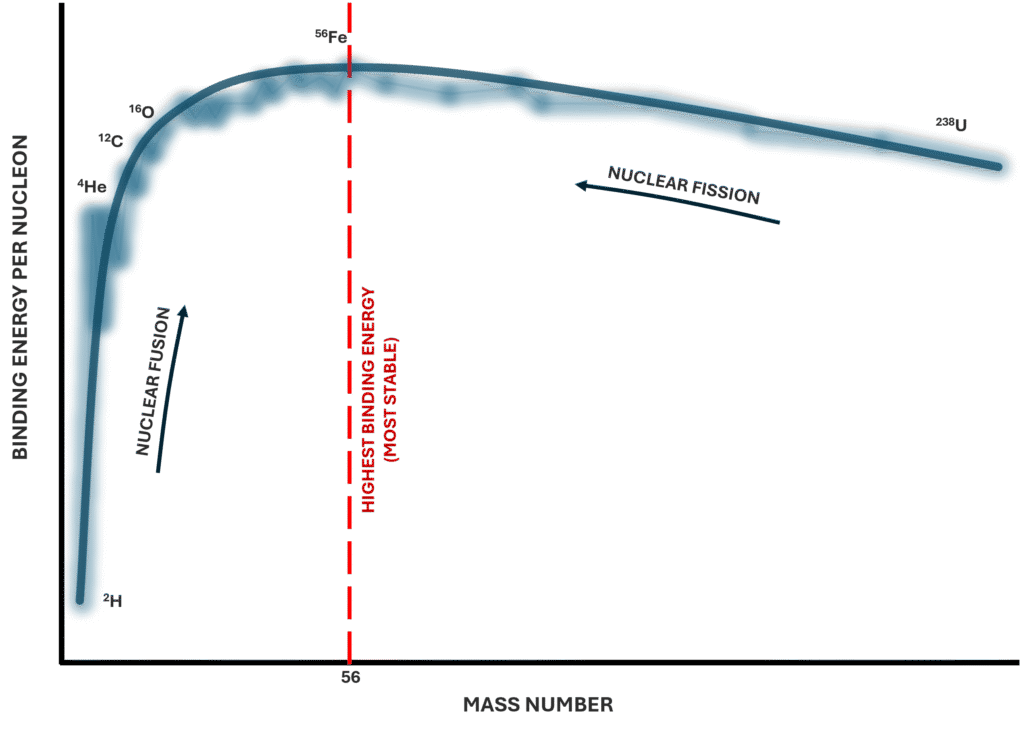
Importantly, binding energy increases as nuclei grow larger, but not in a simple linear way. What matters most for stability is binding energy per nucleon, which compares how tightly each nucleon is bound on average. Nuclei around iron-56 have the highest binding energy per nucleon and are therefore among the most stable.
Very light nuclei have relatively low binding energy per nucleon because they contain too few nucleons to fully benefit from strong-force interactions. Very heavy nuclei also have lower binding energy per nucleon because increasing proton–proton repulsion reduces overall stability. This explains why both very small and very large nuclei are more prone to decay.
Where this matters clinically
Binding energy underlies all radioactive decay processes used in nuclear medicine. Radionuclides decay because doing so moves them toward a configuration with greater binding energy per nucleon. The energy released during decay ultimately produces the gamma photons or positrons detected in imaging.
