What determines nuclear stability?
Nuclear stability is determined by the balance between two opposing forces within the atomic nucleus: the strong nuclear force, which binds protons and neutrons together, and the electrostatic repulsion between positively charged protons, which pushes them apart. A nucleus is stable when the attractive strong force is sufficient to overcome the repulsive forces between protons.
Nuclear stability depends on the balance between the strong nuclear force and proton–proton repulsion, moderated by neutron number and binding energy per nucleon.
The ratio of neutrons to protons (the N/Z ratio) is critical. In light nuclei, stability typically occurs when the number of neutrons and protons is similar. As atomic number increases, additional neutrons are required to offset the increasing electrostatic repulsion between protons. When this balance is disrupted, the nucleus becomes unstable and undergoes radioactive decay to achieve a more stable configuration.
Stability is also influenced by nuclear structure effects, such as pairing of nucleons and so-called “magic numbers,” which correspond to particularly stable arrangements of protons or neutrons.
Understanding the physics
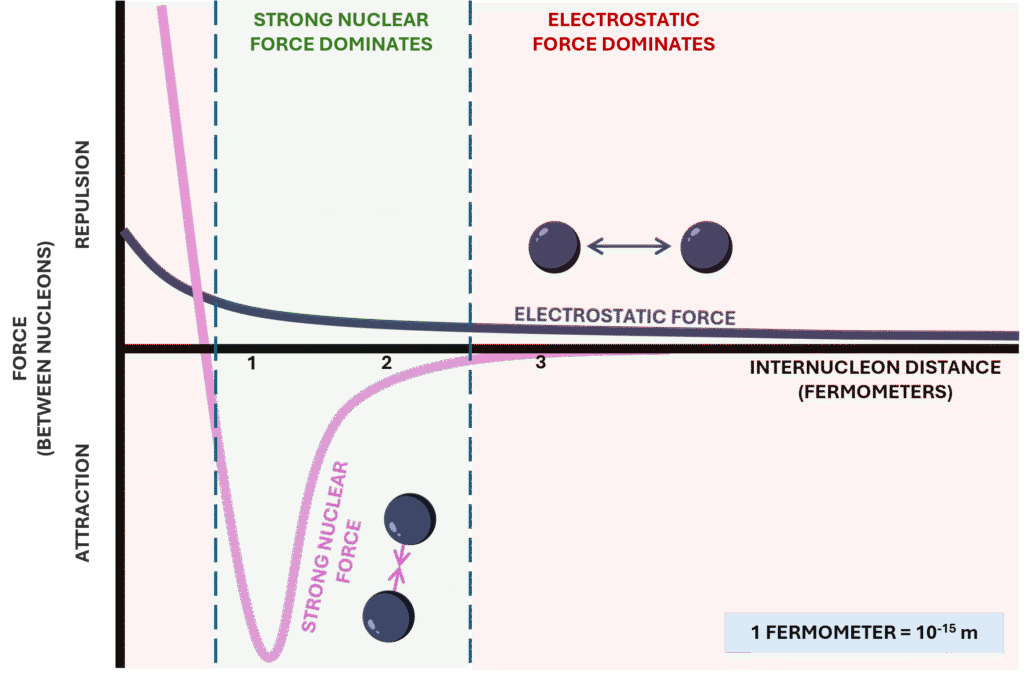
The strong nuclear force is a short-range but extremely powerful attractive force acting between nucleons (protons and neutrons). It operates over distances on the order of a few femtometres and is responsible for binding the nucleus together. Importantly, it acts equally between proton–proton, neutron–neutron, and proton–neutron pairs.
In contrast, electrostatic repulsion acts only between protons and increases as the number of protons increases. Unlike the strong force, the electrostatic force has a longer range and grows with atomic number. As nuclei become larger, the cumulative repulsive force between protons becomes significant.
Neutrons play a stabilising role because they contribute to the strong nuclear force without adding additional electrostatic repulsion. This is why heavier nuclei require a higher proportion of neutrons to remain stable. If a nucleus contains too many neutrons or too many protons relative to the stable range (often visualised as the “band of stability”), it becomes energetically favourable for it to transform via radioactive decay.
Another key determinant of stability is binding energy per nucleon, which reflects how tightly each nucleon is bound within the nucleus. Nuclei with higher binding energy per nucleon are generally more stable. Iron-56 lies near the peak of this curve and is one of the most stable nuclei known. Very heavy nuclei, however, experience such strong proton–proton repulsion that even additional neutrons cannot fully stabilise them, leading to inherent instability.
Even–even nuclei (those with even numbers of protons and neutrons) are typically more stable than odd–odd nuclei due to nucleon pairing effects. Certain “magic numbers” of protons or neutrons correspond to particularly stable nuclear configurations, analogous to closed electron shells in atomic physics.
When the balance between attractive and repulsive forces cannot be maintained, the nucleus reduces its energy through radioactive decay.
| Feature | Strong Nuclear Force | Electrostatic Force (Coulomb Force) |
|---|---|---|
| What it acts between | Nucleons (protons and neutrons) | Electric charges (e.g. proton–proton repulsion) |
| Nature of the force | Attractive (primarily), strong repulsion <0.5 fermometres | Repulsive between like charges, attractive between opposite charges |
| Range | Extremely short (~1–3 femtometres, size of a nucleus) | Long-range (extends infinitely, decreases with distance²) |
| Relative strength | Strongest force in nature (within the nucleus) | Much weaker than the strong force at nuclear distances |
| Distance dependence | Very strong at ~1 fm, rapidly falls to zero beyond a few fm | Follows inverse square law: (F ∝ 1 / r2) |
| Particles involved | Protons and neutrons | Charged particles (e.g. protons, electrons) |
| Effect in nucleus | Holds nucleons together and stabilises nuclei | Causes repulsion between protons, promoting instability |
| Role in nuclear stability | Counteracts proton–proton repulsion | Opposes nuclear binding in proton-rich nuclei |
| Carrier particle (modern physics) | Gluons (via residual strong force between nucleons) | Photons (electromagnetic interaction) |
| Importance in radiology physics | Explains binding energy, nuclear stability, radioactive decay | Explains Coulomb barrier in nuclear reactions and alpha decay |
Where this matters clinically
Understanding nuclear stability explains why only certain isotopes can be used in nuclear medicine. Diagnostic radionuclides must be unstable enough to emit detectable radiation, but sufficiently stable to persist long enough for imaging. Stability principles determine which isotopes exist naturally and which must be produced artificially in reactors or cyclotrons.
Related questions
Why is Tc-99m ideal for radionuclide imaging?
