What is alpha decay?
Alpha decay is a type of radioactive decay in which an unstable nucleus emits an alpha particle, consisting of two protons and two neutrons. This alpha particle is essentially a helium-4 nucleus.
Alpha decay involves emission of a helium nucleus, reducing atomic number by 2 and mass number by 4, and is characteristic of heavy unstable nuclei.
When alpha decay occurs, the parent nucleus loses two protons and two neutrons. As a result, the atomic number decreases by 2 and the mass number decreases by 4. The daughter nucleus is therefore a different element.
Alpha decay occurs primarily in very heavy nuclei, such as uranium and radium.
Understanding the physics
In very heavy nuclei, the balance between the attractive strong nuclear force and the repulsive electrostatic force between protons becomes unstable. As atomic number increases, the repulsive force between positively charged protons becomes significant.
Alpha decay is one way the nucleus can reduce this instability.
An alpha particle (2 protons + 2 neutrons) exists as a tightly bound cluster within heavy nuclei. When emitted, it carries away both mass and positive charge.
The nuclear transformation can be written as:
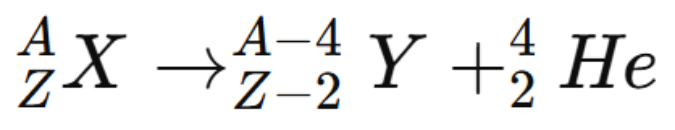
Because alpha particles are relatively large and carry a +2 charge, they interact strongly with matter. They have:
High ionising power
Very short range in tissue (a few micrometres)
Poor penetration (stopped by paper or skin)
For this reason, alpha emitters are not typically used in diagnostic imaging. However, they are increasingly important in targeted radionuclide therapy, where their high ionisation density can destroy tumour cells while limiting penetration into surrounding tissue.
Where this matters clinically
This topic is less common but I want it here for completeness sake.
Alpha decay is not used in SPECT or PET imaging but is highly relevant in radionuclide therapy, particularly with alpha-emitting isotopes used in targeted cancer treatment.
Understanding alpha decay also reinforces the relationship between mass number, atomic number, and nuclear stability which are key principles in radioactive decay.
